

DietGel® Recovery Contains All Six Essential Electrolytes
The body of all animals is made up of approximately 70% water. This number varies amongst species, with 63% for the beagle and 80% for the mouse, for example (1). The body water is contained within 3 major compartments: intracellular (within the cells), interstitial (between the cells), and the blood. How is this balance of fluids maintained in the body? Electrolytes play a vital role in maintaining homeostasis within the body. But, what are electrolytes? What are their roles? And, how can you help make sure that your lab animals get the electrolytes they need in case of dehydration?
Electrolytes – General Functions
Body fluids contain electrolytes, electrically-charged minerals that help regulate osmotic pressure in cells and maintain the function of all the organs, including muscles and nerves. We get them from what we eat and drink. Electrolytes maintain normal fluid levels in the fluid compartments by a process called osmosis: if the electrolyte concentration is high in one compartment, fluid moves into that compartment, and vice versa (2). If the balance of electrolytes is disturbed, disorders can develop and vital body systems can be affected. Electrolytes help regulate muscle, nerve and organ functions, fluid balance and many other biological functions. Electrolyte imbalances are usually associated with fluid loss and dehydration.
For humans, after a hard workout or when recovering from the flu, we are recommended to consume sports drinks such as Gatorade® or Pedialyte®, or chicken broth. Why? You guessed it: because they are high in electrolytes that help your body rehydrate and replenish its osmotic balance faster. In laboratory animals, dehydration can happen during shipment and transportation, surgeries, studies or sickness. DietGel® Recovery with electrolytes can help them recover faster.
To learn more about dehydration causes in lab animals, check out our previous blogs:
Fluid Therapy and Electrolyte Replenishment for Faster Recovery after Surgery
Leading Causes of Dehydration in Laboratory Animals
DietGel® Recovery contains all six essential electrolytes, making it a much more complete formulation compared to other products on the market. For 100g of gel, DietGel® Recovery contains :
- 106 mg of Sodium
- 86 mg of Potassium
- 115 mg of Chloride
- 35 mg of Calcium
- 0.3 mg of Magnesium
- 42 mg of Phosphorus

One cup of DietGel® Recovery, placed at the bottom of a cage and changed every 2-3 days, supports a better and faster recovery, and helps you adhere to the 3Rs for animal welfare standards and recommendations.
REQUEST YOUR FREE SAMPLE OF DIETGEL RECOVERY TODAY!
Six Essential Electrolytes
Sodium (Na+)
Most of the sodium in the body is found outside the cells, in the interstitial compartment or in the blood. Sodium is the major cation in the extracellular fluid. It has a very important role in maintaining blood volume by regulating osmotic pressure. The brain is particularly sensitive to changes in the sodium level in blood. Sodium also has a major role in muscle and nerve functions: sodium channels are located in the membrane of muscle and nerve cells. Mammalian heart cells, for example, typically express more than 100,000 sodium channels!! (3) Sodium channels are responsible for initiation and transmission of electrical impulses through the nerves and muscles by depolarization of the cells. Sodium is thus involved in higher processes such as pain, locomotion and cognition.
Hyponatremia (low level of sodium in the blood), is the most common clinical electrolyte disorder and can cause gait disturbances, cognitive impairments, brain dysfunction and eventually can lead to seizures, coma and death (4). DietGel® Recovery contains 106 mg of Sodium per 100g.
Potassium (K+)
Potassium works hand in hand with sodium. Unlike sodium, potassium is located inside the cells. It is the major cation in the intracellular fluid. Potassium channels are responsible for repolarization of the cells. Cell membranes also contain sodium-potassium pumps that regulate the balance of sodium and potassium intra- and extra- cellularly to maintain cellular volume: when sodium levels go up, potassium levels go down, and vice versa. In the cell membranes, another type of channel, the Na+/K+ pump acts to transport sodium and potassium across the cell membrane in a ratio of 3 sodium ions out for every 2 potassium ions, thereby stabilizing the membrane electric potential (5). This is especially important in neurons, getting them ready to fire electric impulses.
Potassium regulates nerve signals, muscle contractions including cardiac contractions, vascular tone and blood pressure. Consequently, hypokalemia (low potassium level) can cause muscle weakness, cramping, twitching, or even paralyzed, abnormal heart rhythms and high blood pressure. DietGel® Recovery contains 86mg of potassium per 100g.
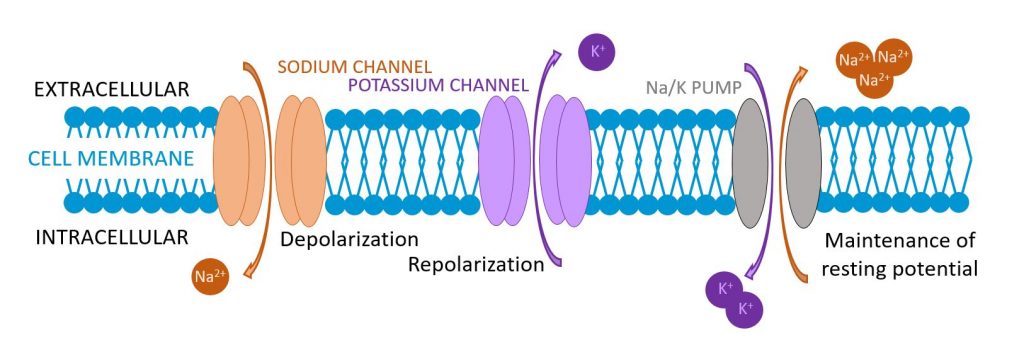
Chloride (Cl-)
Chloride is the most abundant anion in the extracellular fluid. It works with other electrolytes to help regulate the amount of fluid in the body and maintain the acid-base balance, acting as a buffer to maintain electrical neutrality at the cellular level by moving into or out of the cells as needed. There are five main types of chloride channels, involved in a wide range of physiological functions. They are attractive targets for drug development for a wide range of human disorders (6).
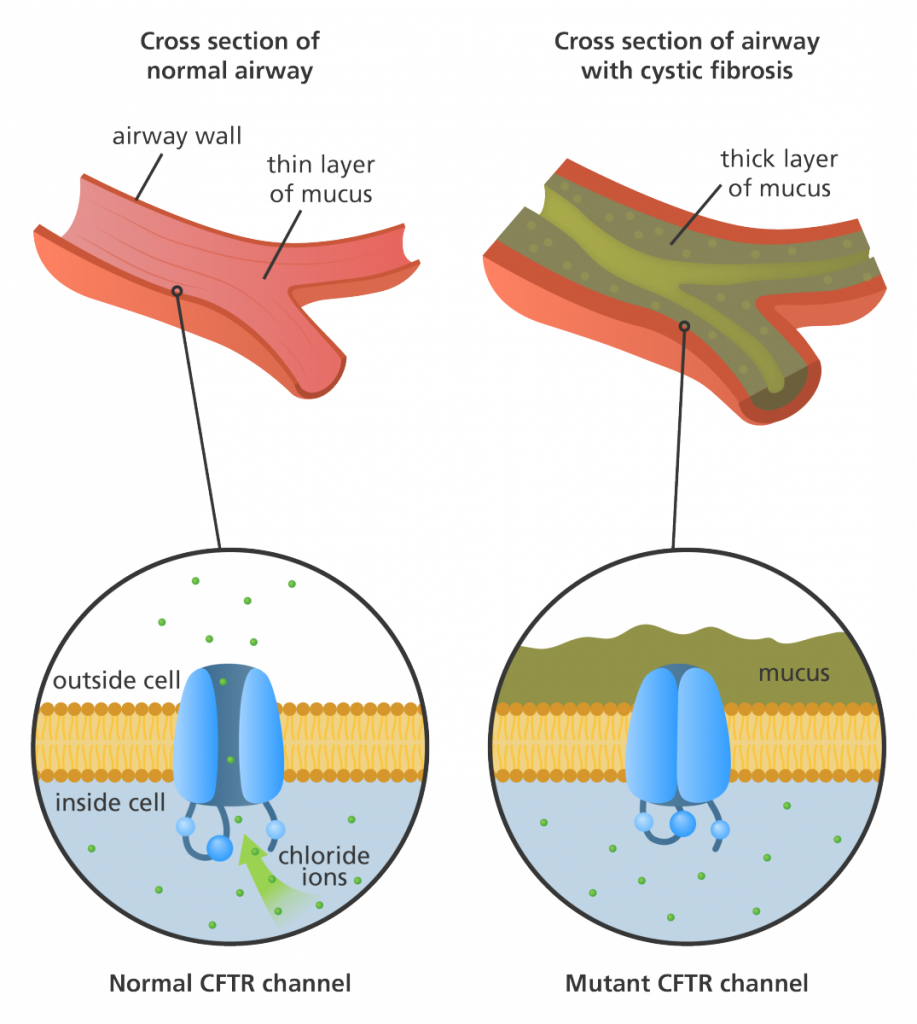
Notably, chloride channels are involved in epithelial fluid secretion, such as saliva, sweat, tears, digestive enzymes, and mucus, a substance that lubricates and protects the lining of the airways, or reproductive system (7 – see image). Dysfunction of the chloride channel CFTR causes disruption of the movement of chloride, leading to thickening and increased stickiness of secretions throughout the body, and causing Cystic Fibrosis (8). In the nervous system, chloride channels are involved in processes including the modulation of the excitability of neurons, skeletal, cardiac and smooth muscles, neuronal growth, development and neurotransmitter uptake (9).
Signs of chloride deficiency (hypochloremia) are poor growth, apathy, irregular heart rate and muscle stiffness and cramps.
DietGel® Recovery contains 115mg of chloride per 100g gel.
Calcium (Ca2+)
Almost all calcium is stored in bones and teeth. The body moves calcium out of bones into blood as needed to maintain a steady level of calcium in the blood. Calcium is regulated and maintained by 3 components:
- Vitamin D that helps the absorption of calcium through the GI tract
- Parathyroid hormone which stimulates bones to release calcium into blood
- Calcitonin which has the reverse effect, slowing the breakdown of bone
Calcium is of course essential for the formation of bone and teeth, but also for neurotransmission, muscle contraction, blood clotting, normal heart rhythm, regulation of enzymes and hormone secretion.
In case of hypocalcemia (lack of calcium in the blood stream), calcium is pulled out from the bone into the blood stream, through bone breakdown and resorption, and osteoporosis can result. As calcium is also highly involved in muscle and nerve function, hypocalcemia can cause muscle spasms and tetany and lead to respiratory failure. DietGel® Recovery contains 35 mg of calcium per 100g gel.
Magnesium (Mg2+)
Similar to calcium, magnesium is necessary for the formation of bone and teeth and for normal nerve and muscle function. It is also involved in over 300 enzyme systems that regulate diverse biochemical reactions, including including protein synthesis, blood glucose control, and blood pressure regulation (10). About half of the magnesium is found in bone and teeth, the rest is in soft tissues, and less than 1% is in blood. Hypomagnesemia (lack of magnesium) contributes to osteoporosis. It also causes weakness, muscle spasms, tremors, and seizures. DietGel® Recovery contains 0.3 mg of magnesium per 100g of gel.
Phosphorus/Phosphate (P/PO43-)
In the body, almost all phosphorus is combined with oxygen, forming phosphate. This mineral is an essential component mostly found in bones and teeth. The remainder is located primarily inside the cells, where it is involved in energy production as phosphate compounds, such as adenosine triphosphates (ATP), the body’s source of biochemical energy. Phosphate is also a primordial component of the DNA backbone. Phosphorus is involved in many physiologic functions, including cardiac and diaphragmatic muscular function. Hypophosphatemia can lead to loss of appetite, muscle impairment, bone pain and fractures, tooth decay, decreased cardiac activity and irregular heartbeat (11). DietGel® Recovery contains 42mg of phosphorus per 100g of gel.
Electrolytes are involved in so many bodily functions. If electrolyte levels are too low, cell and organ function will decline, leading to health and welfare issues in your laboratory animals. Make sure you notice the signs of dehydration, and provide hydration with electrolytes to help their recovery!
REQUEST YOUR FREE SAMPLE OF DIETGEL RECOVERY TODAY!
References:
(1) A review of body composition studies with emphasis on total body water and fat
(2) Merck – Electrolytes Balance
(3) Structure and function of voltage-gated sodium channels
(4) Chronic Hyponatremia Causes Neurologic and Psychologic Impairments
(5) Sodium-Potassium Pump
(6) Chloride channels as drug targets
(7) What is cystic fibrosis?
(8) Cystic Fibrosis (CF)
(9) Chloride Homeostasis in Neurons With Special Emphasis on the Olivocerebellar System: Differential Roles for Transporters and Channels
(10) Magnesium Fact Sheet – NIH
(11) Electrolyte series – Calcium and phosphorus
Additional references:
(12) What are electrolytes (ions)?
(13) Electrolytes

